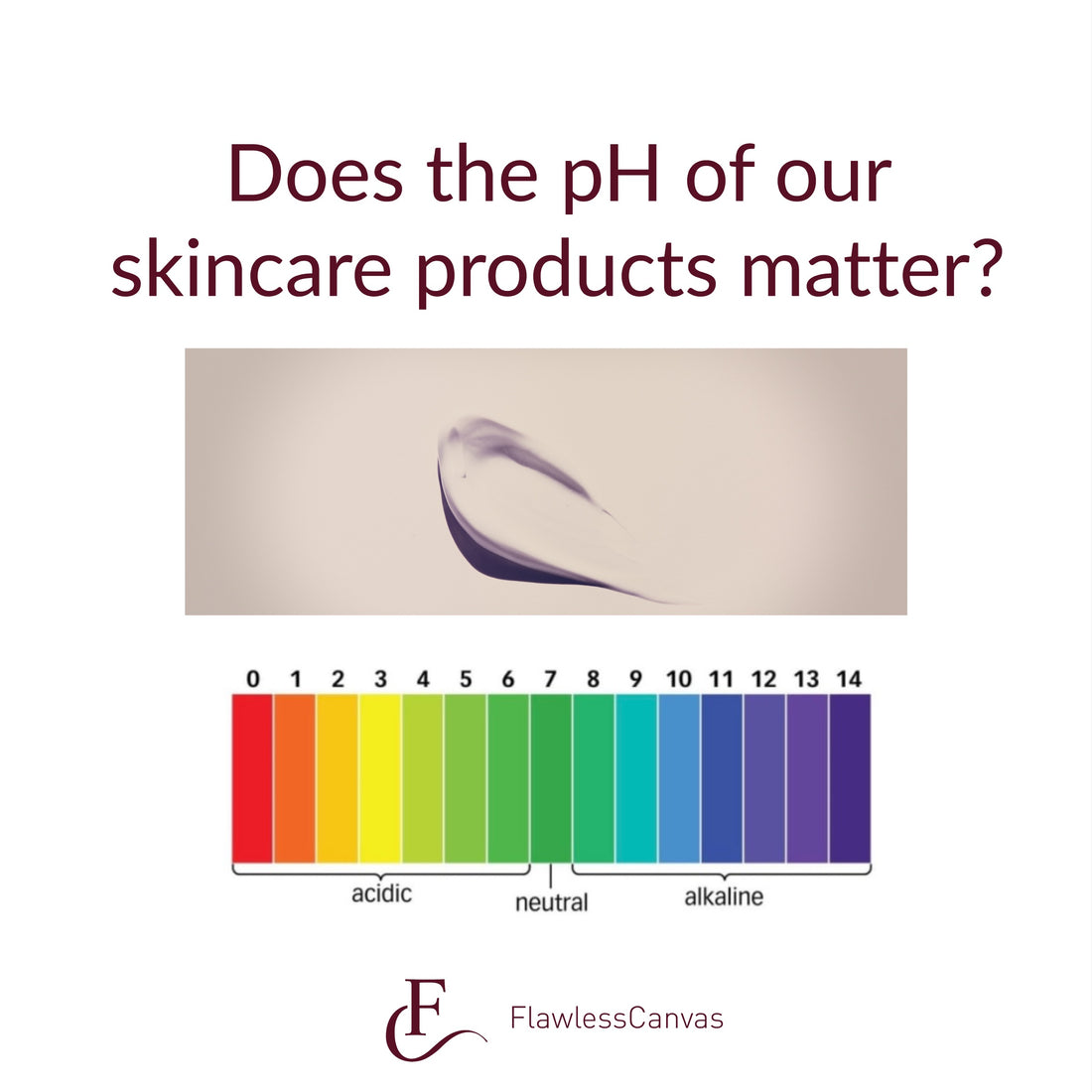
In 1928 Schade and Marchionini first coined the term Säuremantel or acid mantle, which described the inherent acidic nature of the stratum corneum (pH 4.3-5.3). SC pH is critical to preserve barrier function determining cohesion/ integrity and preserving the microbiota. It is logical to assume that skincare formulations should be acidic. Skin friendly pH formulations are especially important for subjects affected by skin diseases or for older individuals, as the skin surface pH becomes alkaline under these circumstances.
Does the pH of your skincare matter? Yes, although it is important to remember that the skin has a self-neutralizing capacity. What does this mean? It means the skin has mechanisms to restore the pH to the correct levels. This can take from minutes to hours depending on the severity of the change. For example, washing hands with soap bars increases skin pH in about 3 units for at least 90 min. Small and sustained pH increases triggered by the daily use of skincare products with higher pH triggers skin barrier dysfunction with decreased repair mechanisms resulting in skin dehydration. Hence, I prefer to use skin friendly pH formulations. Not convinced yet? Here are some examples that support the importance of using skincare with skin friendly pH:
(1) Polyhydroxy acids produce skin acidification that is linked to enhanced barrier function (this has been demonstrated in mice and humans).
(2) Lactic acid (4%, pH 3.7-4.0) improves barrier function, increases ceramide production and reduces the sensitivity to SLS.
(3) Acidic electrolyte water (pH 2.0-2.7) reduces dermatitis severity and S.aureus colonization.
(4) A healthy skin microbiome is observed at acidic pH. Pathogenic bacteria such as S.aureus thrive at neutral pH. What does this mean? It means applying skincare with an abnormally high pH may tilt the balance toward pathogenic bacterias growth resulting in skin complication. Remember S.aureus is already a resident in our skin BUT is kept under control due to the acidic pH and all the antibacterial peptides produced by our “good” acidic pH loving bacteria.
(5) P.acnes grows better at pH 6.0-6.5
(6) Subjects prone to irritant contact dermatitis have higher skin pH.
(7) Enzymes involved in skin desquamation work in a pH depending matter. Thus, an alkaline pH disturbs normal skin desquamation.
And here is a final example: let’s talk about niacinamide, one of the most Googled active ingredients of 2020. Traditionally, formulations containing niacinamide tend to have neutral pH to prevent the formation of niacin, a vasodilator responsible for flushing (redness linked to some products containing niacinamide). Interestingly, niacinamide at 2% can be formulated into acidic products (as low as pH 3.8) with minimal niacin formation. This is the reason why all my products are skin friendly pH formulations containing 2% niacinamide: all benefits, minimal complications. Acidic formulations of niacinamide have increased clinical efficacy as they activate the process by which cells get rid of the damaged structures that accumulate preventing their proper function (AKA autophagy) .
FUN FACTS : (1) Did you know that newborn babies have a skin pH of 7.08? Yes, we are born with higher skin pH which rapidly decreases to the acidic levels (5.7) within the first few days of being born. (2) Common skin conditions in the newborn such as diaper dermatitis and eczema are linked to higher pH values.
#skincarescience #Flawlesscanvas_skincare #theskinbiologist #skinph #antiaging #skincareproduct #skincare #skinhealth #skincarejunkie #beautyscience